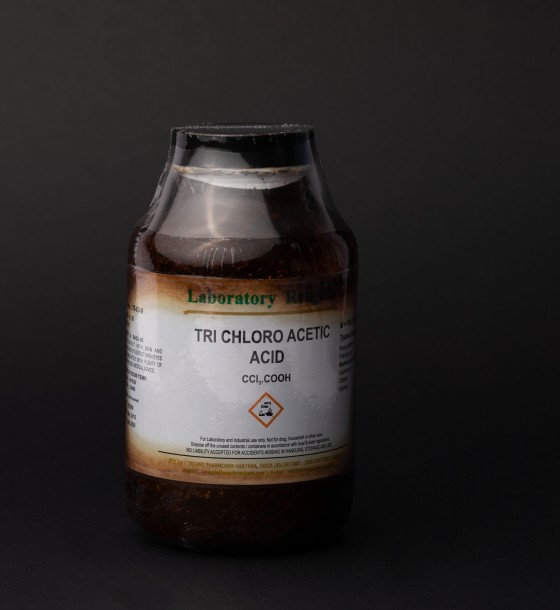
TRIchloroacetic acid
234.00
In Stock
Trichloroacetic acid (TCA) is a strong, corrosive organic acid commonly used in various chemical, laboratory, and medical applications. Its chemical formula is C₂HCl₃O₂. It is known for its powerful ability to break down proteins and is widely used for skin treatments, research, and in analytical chemistry.
Key Properties:
- Appearance: White crystalline solid or colorless liquid.
- Odor: Sharp, pungent odor.
- Solubility: It is soluble in water, alcohol, and acetone.
- Corrosive: Trichloroacetic acid is highly corrosive and can cause severe skin burns and eye damage.
Common Uses:
Medical Applications:
- Chemical Peels: TCA is often used in dermatology for chemical peeling procedures. It helps remove the outer layers of the skin to treat conditions like acne scars, age spots, and wrinkles.
- Wart Removal: It is used to treat certain types of warts, especially genital warts, by causing tissue destruction at the site of application.
Laboratory Applications:
- Protein Precipitation: TCA is commonly used to precipitate proteins from a solution in biochemical and molecular biology experiments.
- DNA and RNA Extraction: It helps in the extraction and purification of nucleic acids, by precipitating proteins and other contaminants from biological samples.
Analytical Chemistry:
- Acidic Medium: TCA is used in titrations and as a reagent in certain chemical analyses due to its ability to create an acidic environment.
Other Uses:
- Industrial Use: It may also be used in some industrial applications, such as for the removal of impurities from certain chemical processes.
Safety Considerations:
- Protective Equipment: Because of its corrosive nature, it’s important to wear gloves, eye protection, and a lab coat when handling TCA.
- Ventilation: Use in a well-ventilated area to avoid inhaling fumes.
Trichloroacetic acid (TCA) is a strong, corrosive organic acid commonly used in various chemical, laboratory, and medical applications. Its chemical formula is C₂HCl₃O₂. It is known for its powerful ability to break down proteins and is widely used for skin treatments, research, and in analytical chemistry.
Key Properties:
- Appearance: White crystalline solid or colorless liquid.
- Odor: Sharp, pungent odor.
- Solubility: It is soluble in water, alcohol, and acetone.
- Corrosive: Trichloroacetic acid is highly corrosive and can cause severe skin burns and eye damage.
Common Uses:
Medical Applications:
- Chemical Peels: TCA is often used in dermatology for chemical peeling procedures. It helps remove the outer layers of the skin to treat conditions like acne scars, age spots, and wrinkles.
- Wart Removal: It is used to treat certain types of warts, especially genital warts, by causing tissue destruction at the site of application.
Laboratory Applications:
- Protein Precipitation: TCA is commonly used to precipitate proteins from a solution in biochemical and molecular biology experiments.
- DNA and RNA Extraction: It helps in the extraction and purification of nucleic acids, by precipitating proteins and other contaminants from biological samples.
Analytical Chemistry:
- Acidic Medium: TCA is used in titrations and as a reagent in certain chemical analyses due to its ability to create an acidic environment.
Other Uses:
- Industrial Use: It may also be used in some industrial applications, such as for the removal of impurities from certain chemical processes.
Safety Considerations:
- Protective Equipment: Because of its corrosive nature, it’s important to wear gloves, eye protection, and a lab coat when handling TCA.
- Ventilation: Use in a well-ventilated area to avoid inhaling fumes.
0 Reviews for TRIchloroacetic acid